Content
All issues / Volume 18 (2024) / Issue 3 (March)

This is an editorial article. It has no abstract.
Asymmetric polyethersulfone membranes modified with polyelectrolyte complexes: Morphology, thermal analysis and transport properties
Rayane da Silva Vale, Vanessa Bezerra da Silva, Jonas Contiero, Daniel M. Knauss, Caio Marcio Paranhos
Vol. 18., No.3., Pages 245-259, 2024
DOI: 10.3144/expresspolymlett.2024.18
Vol. 18., No.3., Pages 245-259, 2024
DOI: 10.3144/expresspolymlett.2024.18
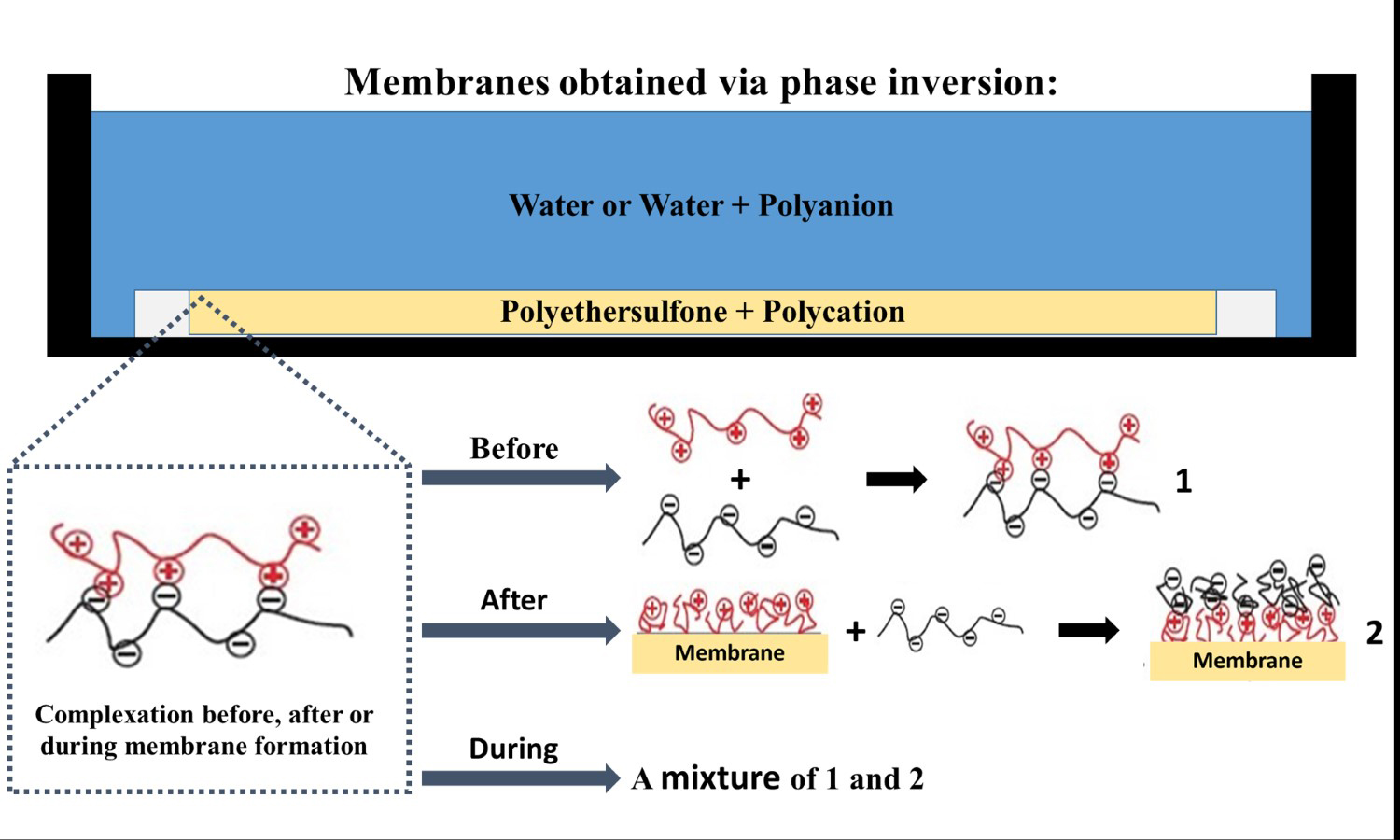
Mixing additives into a polymeric membrane constitutes an easy way to change their properties. Polyelectrolyte complexes (PECs) (polyelectrolytes combined via electrostatic interaction) are good materials for this due to their hydrophilicity, tunable surface charge, and stable structure. In the present study, membranes containing PECs were obtained using three methodologies: complexation before, after, and during membrane formation. The chosen polyelectrolytes were 4-fluorophenyl sulfone-terminated poly(diallylpiperidinium hexafluorophosphate) and sulfonated poly(ethylene terephthalate); the polymeric matrix was polyethersulfone. Fourier transform infrared spectroscopy confirmed the presence of PECs in the membranes. Scanning electron microscopy images of the membranes showed a dense skin and porous sub-layer, with variations in the thickness of the dense layer influenced by the presence of polyelectrolytes. The increase in water vapor permeability indicated that the complexes alter the hydrophilic profile of the membranes. The thermogravimetric curves showed different thermal behaviors depending on the interaction between the polyelectrolytes. All membranes demonstrated enhanced retention of Remazol red dye during filtration, with retention rates exceeding 95%. Notably, the Blend membrane exhibited the highest retention (over 98%) of the dye.
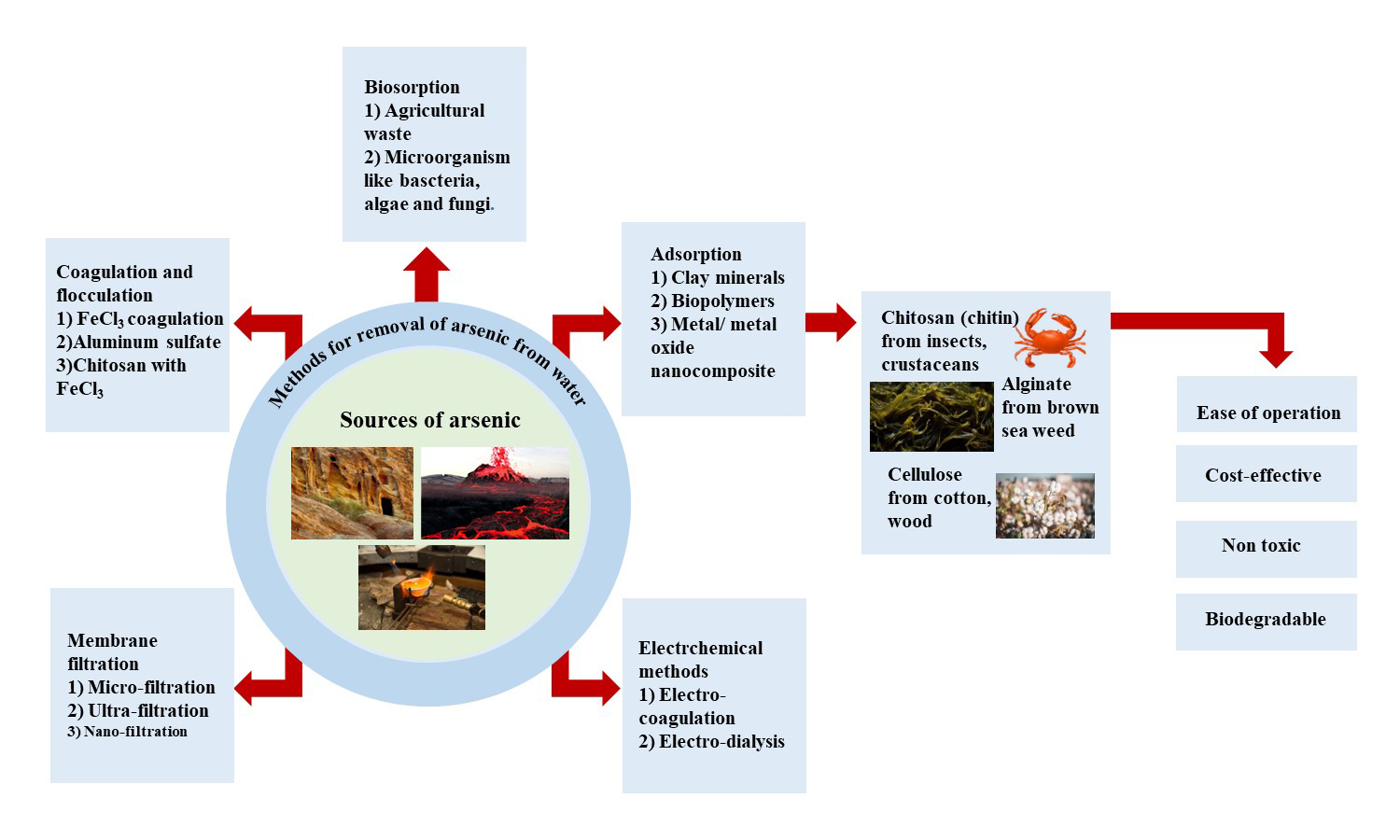
Arsenic, an element found in the Earth’s mantle, can be highly toxic, especially in its As (III) form. It enters our food chain through human activities like melting metals, using arsenic-based pesticides, and natural processes like volcanoes and rock breakdown. Consuming too much arsenic is extremely dangerous, impacting many countries worldwide. To tackle this issue, various methods like filtering, adding chemicals, and using electricity have been developed to clean arsenic-contaminated water. Among these, adsorption is a standout approach due to its simplicity and effectiveness. Biopolymers from living sources offer a natural solution, easily tweaked for arsenic removal. These biopolymers contain functionalities that can strongly latch onto toxic materials, acting like magnets. By customizing them with compounds like titanium dioxide (TiO2), magnetite (Fe3O4), and others, they become even better at capturing arsenic, shaped into tiny particles or beads. This adaptation makes biopolymers a promising choice for cleaning arsenic from water. This review focuses on ways to clean water, specifically exploring how materials like chitosan, alginate, and modified cellulose can be used to remove arsenic by adsorption. It investigates how these materials work under different conditions, highlighting important details. By sharing these insights, this article contributes to the ongoing efforts to ensure cleaner water resources.
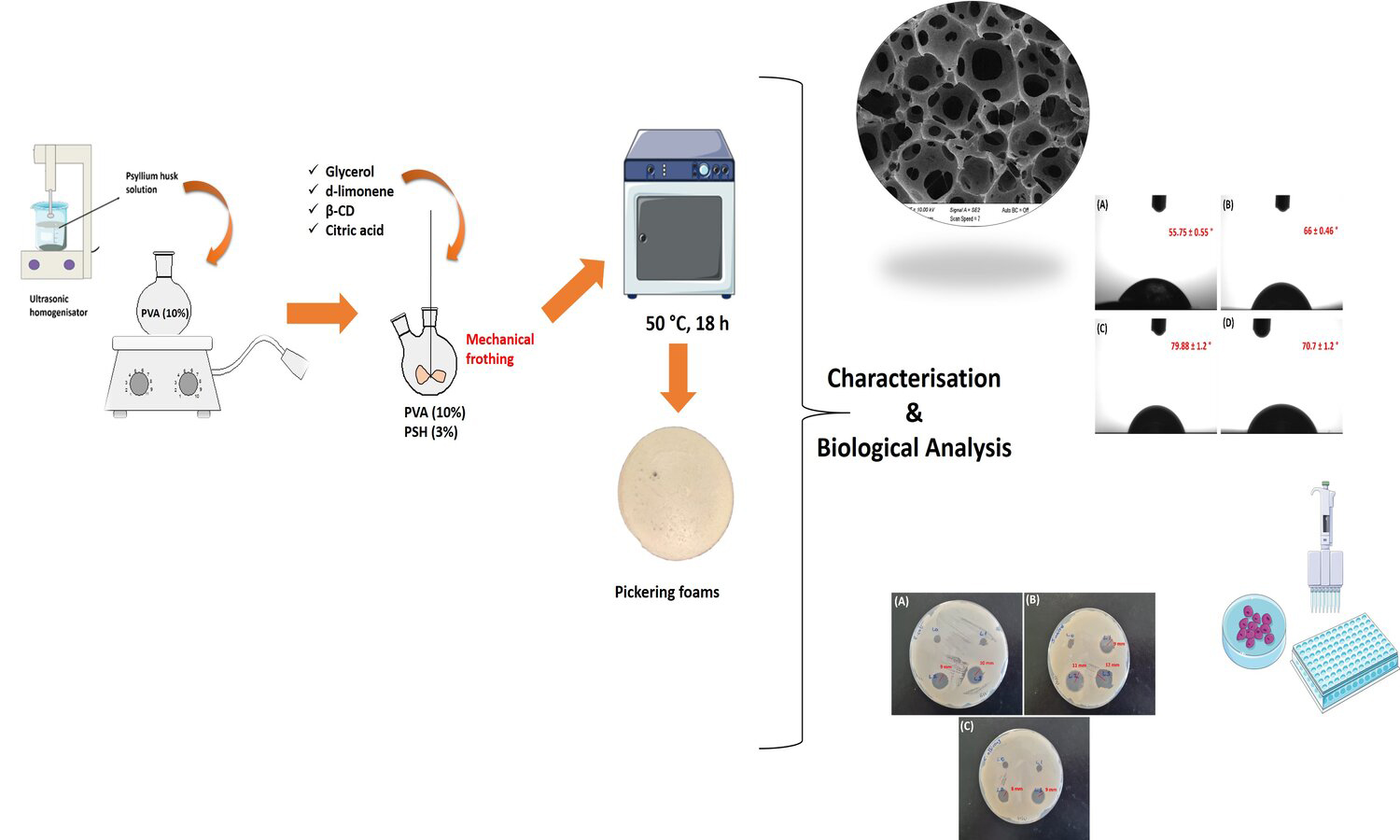
In this study, Pickering foamed emulsions have been prepared using β-cyclodextrin (β-CD), and d-limonene as a surfactant and an oil phase, respectively. The incorporation of β-CD/d-limonene inclusion complexes (ICs) in specific proportions (1:1, 1:3, and 1:5) to water phase, which is a polymer matrix composed of a mixture of polyvinyl alcohol (PVA) and psyllium husk (PSH) by mechanical frothing at high speed, and air bubbles have been formed in oil in water (o/w) emulsions. Ecofriendly bio-based foams have been developed in this method. Scanning Electron Microscope (SEM) analysis showed PVA/PSH Pickering foams usually open porous morphologies and the addition of d-limonene increases the amount of porosity from 43 to 49%. Although the resulting foams indicated similar thermal degradation profile, the presence of d-limonene in foams increased thermal stability. The surfaces of foams have a hydrophilic property with contact angles values lower than 80°. The tensile strength of foams decreased from 170 to 100 kPa due to the increased porosity. All foams indicated antibacterial activity to Staphylococcus aureus (S. aureus) with 9–12 mm zone inhibition. The incorporation of d-limonene into foams surprisingly decreased the cell viability. In brief, our findings show that the Pickering foams can be beneficial for wound healing applications.
Studies of the in vivo bioresorption rate of composite filaments on the basis of polylactide filled with chitin nanofibrils or silver nanoparticles
Konstantin Vadimovich Malafeev, Olga Andreevna Moskalyuk, Vladimir Evgenyevich Yudin, Dmitry Nikolayevich Suslov, Elena Nikolaevna Popova, Elena Mikhaylovna Ivan’kova, Alena Alexandrovna Popova
Vol. 18., No.3., Pages 296-308, 2024
DOI: 10.3144/expresspolymlett.2024.21
Vol. 18., No.3., Pages 296-308, 2024
DOI: 10.3144/expresspolymlett.2024.21
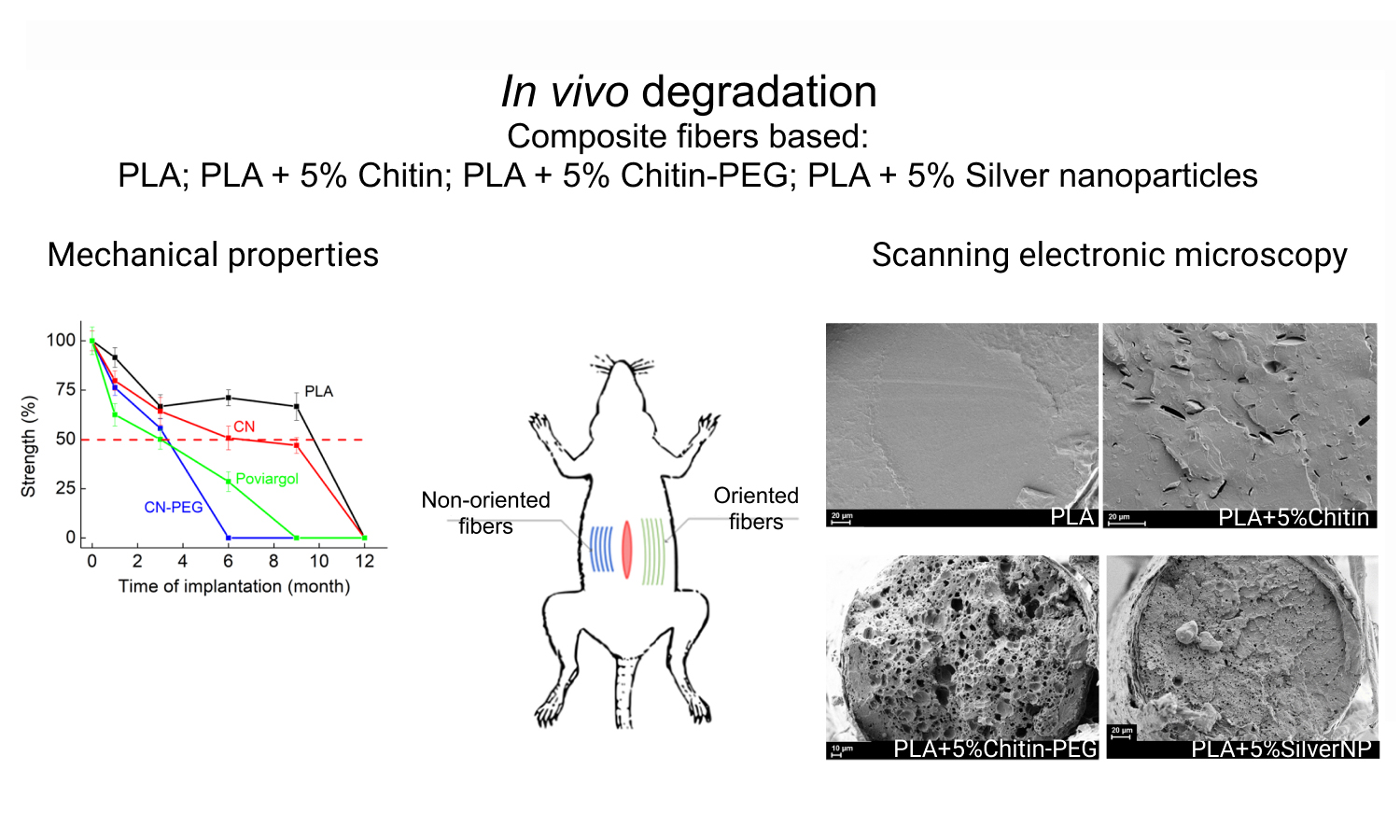
The rates of in vivo bioresorption of composite monofilaments based on polylactide (PLA) containing chitin nanofibrils of two types (pure chitin (CN) or chitin modified with poly(ethylene glycol) (CN-PEG)) or silver nanoparticles stabilized with poly(N-vinylpyrrolidone) (Poviargol) were studied. The in vivo bioresorption rate and its dependence on the degree of orientational drawing of the samples (which varied from 1 in non-oriented samples to 4 in oriented samples) were investigated up to 12 months after implantation. Bioresorption of the samples was monitored using differential scanning calorimetry, scanning electron microscopy, and mechanical tests. Using the differential scanning calorimetry (DSC) method, it was shown that there is a gradual decrease in molecular weight due to a decrease in the temperatures of phase transitions and changes in peak shapes. It has also been shown that the addition of fillers containing water-soluble polymers accelerates the bioresorption of composite sutures. A thread made from pure PLA lost half its strength by the 9th month of implantation, whereas for threads with the addition of CNPEG or Poviargol, this happened after 3.5 months. This causes leaching of water-soluble agents and changes in the supramolecular structure of the filament. This study shows the promise of using these composite threads as a suture material.
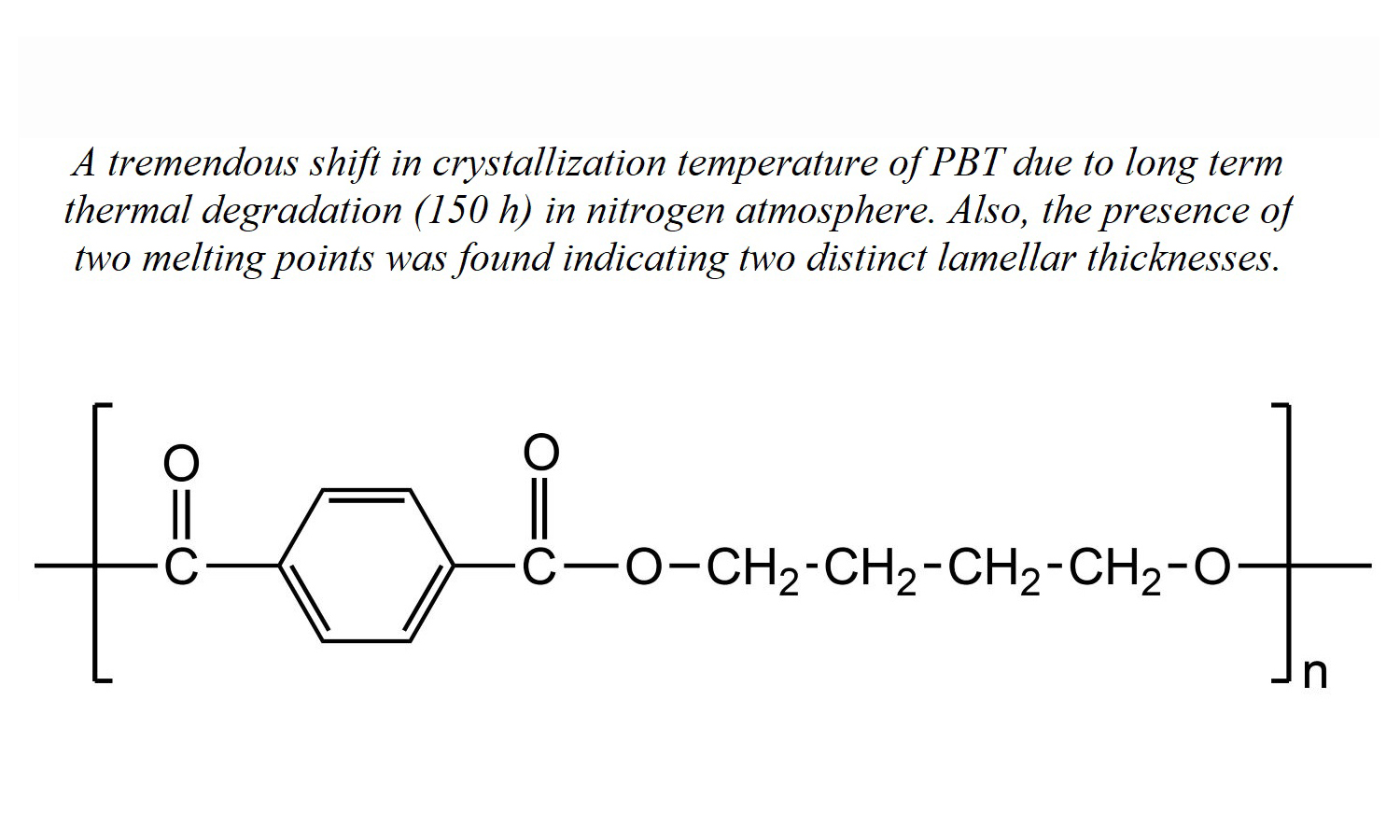
Our work reveals a notable shift in the crystallization temperature (Tc) of poly(butylene terephthalate) (PBT) at which crystallization occurs due to exposure to prolonged thermal degradation at 270°C in an environment of nitrogen gas. The initial Tc of 193°C undergoes a marked decrease, settling at 133°C, which signifies a considerable 60°C shift towards lower temperature ranges. This transition is discernible across three distinct degradation stages: an initial phase of increase, an intermediate phase characterized by a sharp decline, and a subsequent late stage of the degradation phase characterized by a more moderate decrease in Tc. Both crystallinity and crystallization kinetics consistently mirror this pattern, demonstrating an initial rise, a rapid subsequent drop, and a gradual decline in the late-stage period. Evident from the presence of two melting peaks, the research implies differing lamellar thicknesses. As the degradation progresses, the melting points of these peaks, denoted as Tm1 and Tm2, decline at 38 and 41°C, respectively. Validation of the degradation-induced changes is provided by a small angle X-ray scattering (SAXS), which corroborates the observed decrease in the long period (L). A contextualization of the results against prior studies underscores analogous trends in the alteration of crystallization behaviour consequent to degradation.
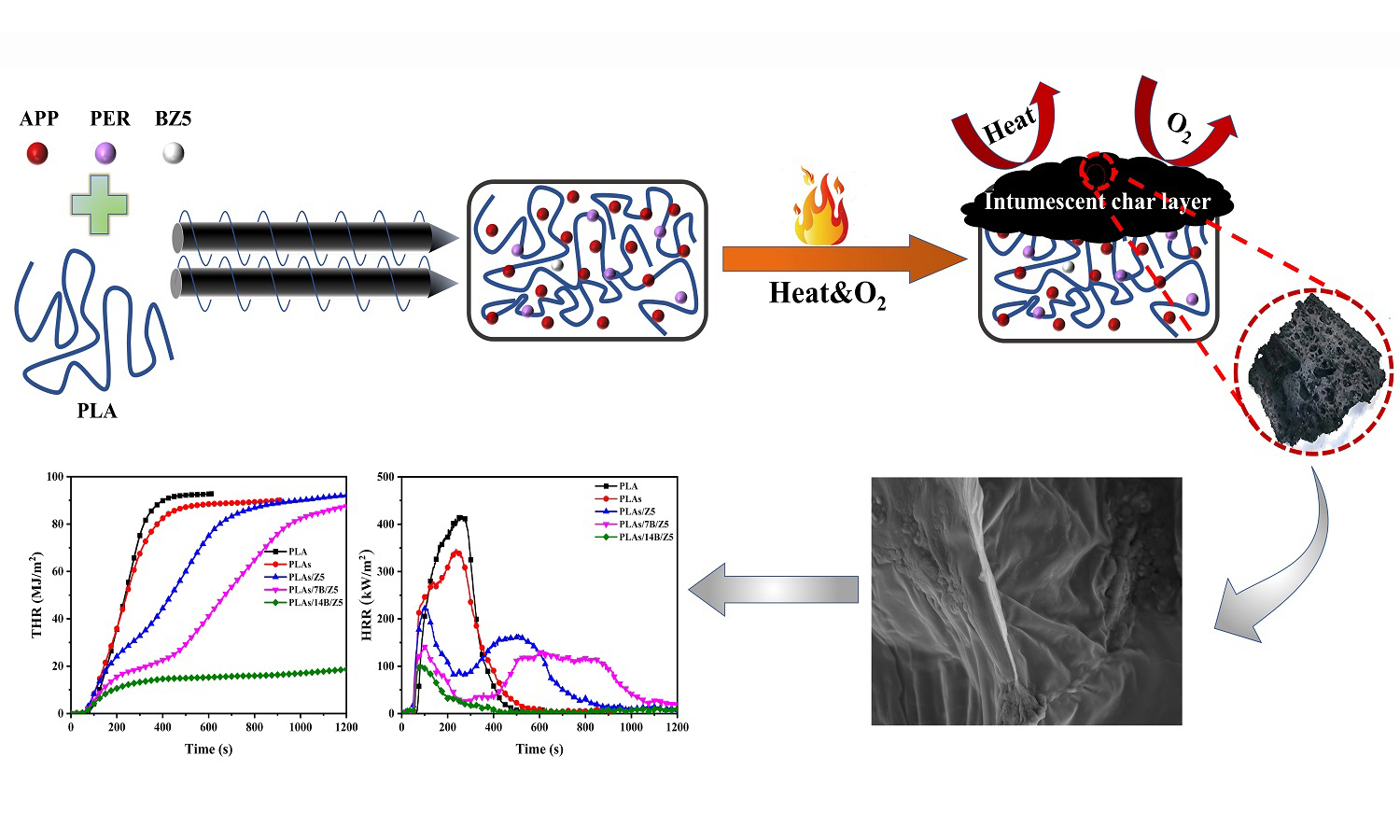
In order to improve the inherent flammability of polylactic acid (PLA) and make this new biodegradable material more widely used, B-modified ZSM-5 (B/Z5) was designed as synergistic agent of intumescent flame retardant (IFR), composed of ammonium polyphosphate (APP) and pentaerythritol (PER), and introduced into PLA/IFR (PLAs) to fabricate PLAs/B/Z5 composites by melt extrusion. The flame retardancy, surface morphology, and thermal stability of the composites were investigated by TG, LOI, UL-94, CCT, and SEM. The addition of B/Z5 makes the decomposition temperature of T5% as well as T10% of PLAs reduction in nitrogen atmosphere and carbon layer form in advance. UL-94 test shows the composites reach V-0 rating. LOI value also steadily increases, and the maximum value is 36.0% with B actual loading of 5.26% (recoded as 14B/Z5) in B/Z5. The PHRR decreases by 75% from 417.77 kW/m2 for pure PLA to 103.19 kW/m2 for PLAs/14B/Z5, and THR reduces from 92.78 to 19.26 MJ/m2, which decreases by about 80% and the char yield reaches 50%. This study provides a simple and green method for the preparation of high flame-retardant PLA, which has a broad practical application prospect.
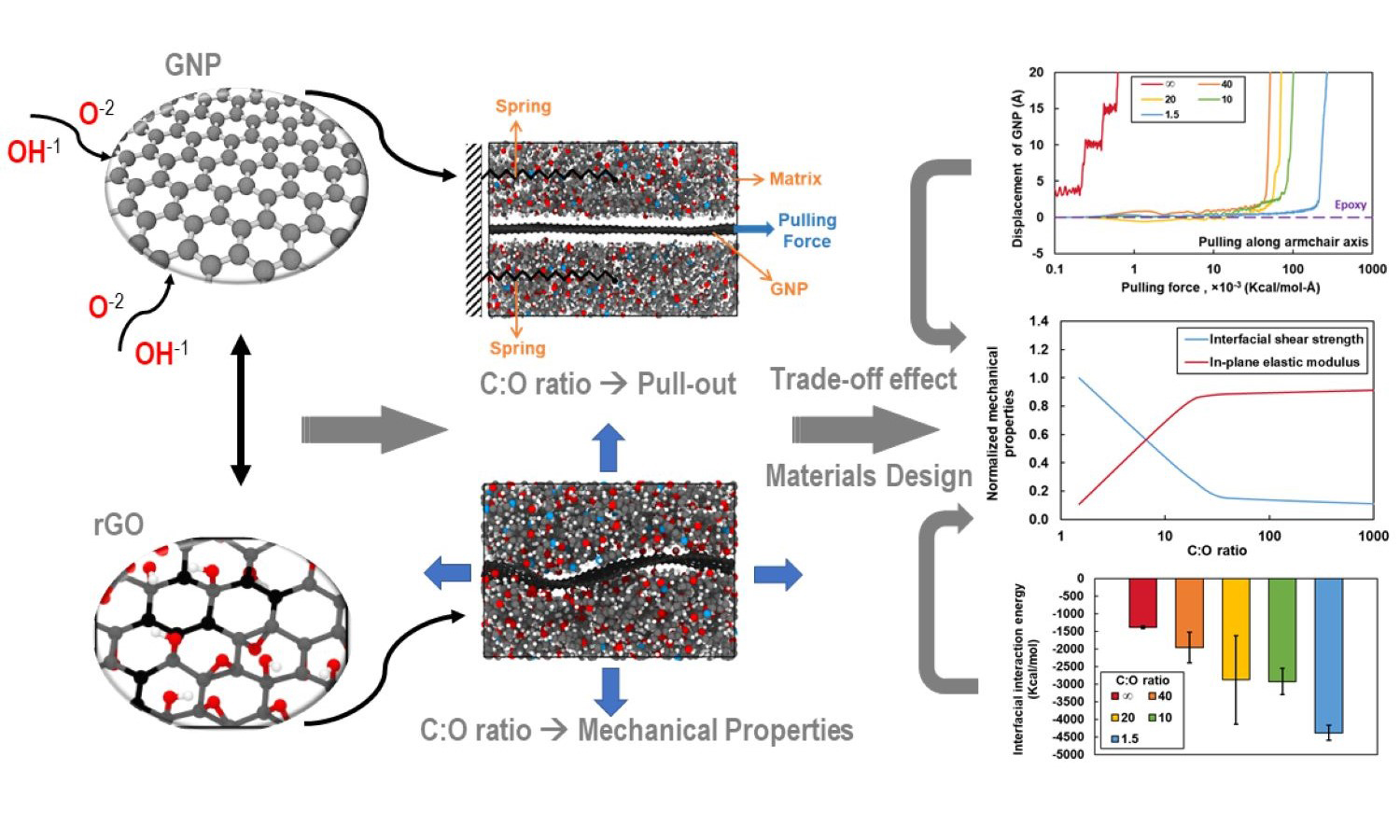
Graphene oxide (GO) nanoplatelets can be used to reinforce neat resin or the matrix phase of fiber composites for improved mechanical properties. Although the oxygen content levels in GO and reduced graphene oxide (rGO) strongly influence the interface with the polymer, experimental-based optimization of the oxygen content for enhanced composite properties is difficult and time-consuming. Fortunately, molecular dynamics (MD) simulation can be used to efficiently predict the interfacial properties of composite on the molecular level and provide physical insight into the effect of the oxygen content of rGO. In this study, MD is used to predict the elastic properties of rGO/epoxy interfaces and the corresponding interfacial shear strength (IFSS) for different oxygen content levels. The results indicate that increasing levels of oxygen in rGO results in interfaces with a reduced in-plane elastic modulus but a substantially higher IFSS. These results are important for the design of rGO/epoxy composites for specific engineering applications.


