Content
All issues / Volume 17 (2023) / Issue 12 (December)

This is an editorial article. It has no abstract.
Construction of a thermally conductive network to improve the thermal and mechanical performance of silicone rubber foam
Hongjie Xie, Lijuan Zhao, Yanli Chen, Bing Han, Yu Hua, Dongliang Zhang, Zhaoqiang Li, Qibo Deng, Yunfeng Zhao
Vol. 17., No.12., Pages 1182-1199, 2023
DOI: 10.3144/expresspolymlett.2023.90
Vol. 17., No.12., Pages 1182-1199, 2023
DOI: 10.3144/expresspolymlett.2023.90
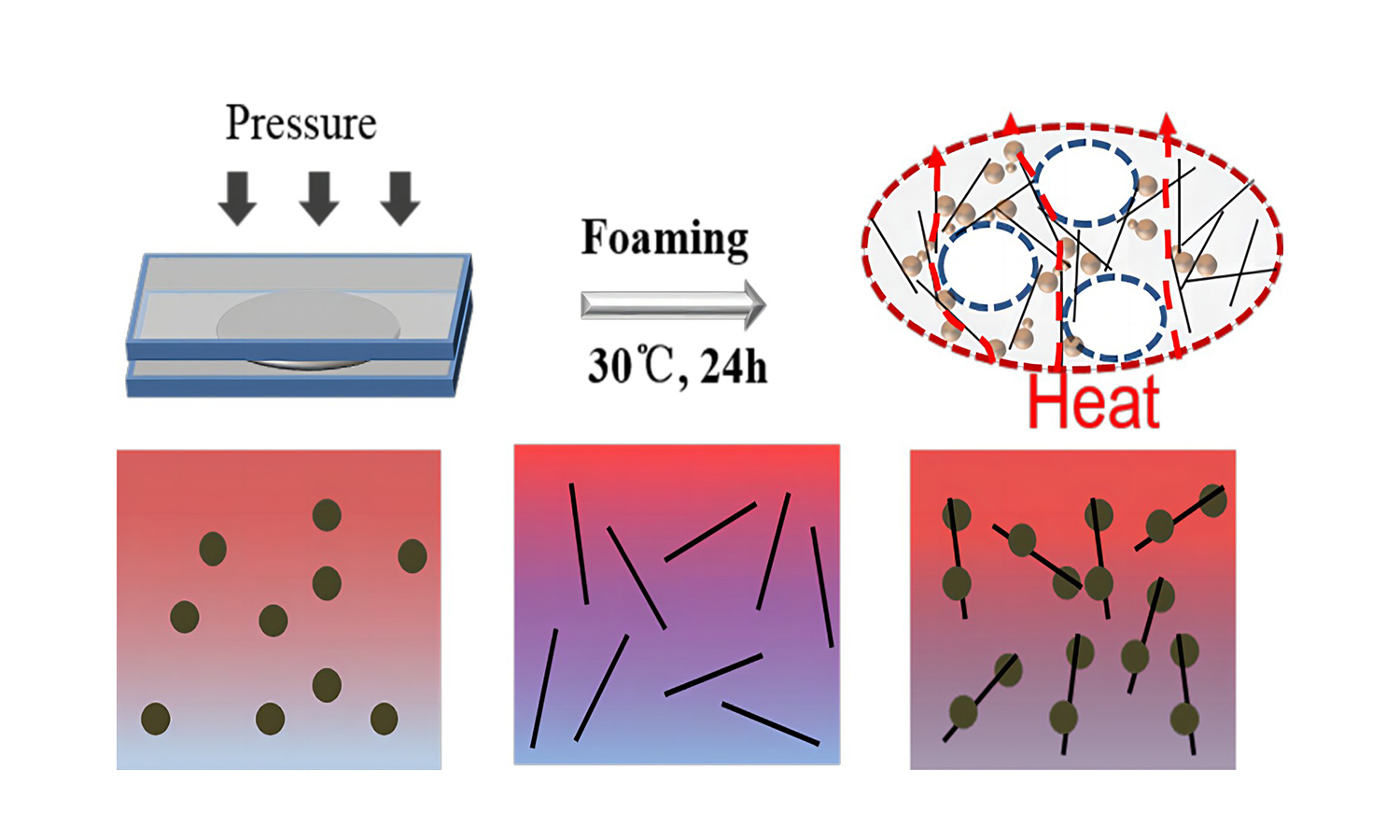
Silicone foam (SF) is a porous silicone rubber with a lower density, higher elasticity, and good thermal stability. In this work, we selected aluminum spheres and carbon fiber (CF) as thermally conductive fillers to prepare hybrid SF. After optimization, we found that Al and CF hybrid SF (Al-CF-SF) has a higher thermal conductivity (1.37 W·m–1·K–1) than the single-filler filled SF (CF-SF, 1.2 W·m–1·K–1 or Al-SF, 0.52 W·m–1·K–1) under the same filling amount of 60 wt%. The finite element simulation was used further to explore the thermal conductive mechanism of the hybrid SF. Meanwhile, the compressive and tensile modulus of the material (CF-SF) was increased to 10.8 and 3.3 MPa compared with pure SF, respectively, and the mechanical properties were improved. In addition, infrared thermography further demonstrated that Al-CF-SF has a faster heat transfer rate under relaxation and applied pressure.
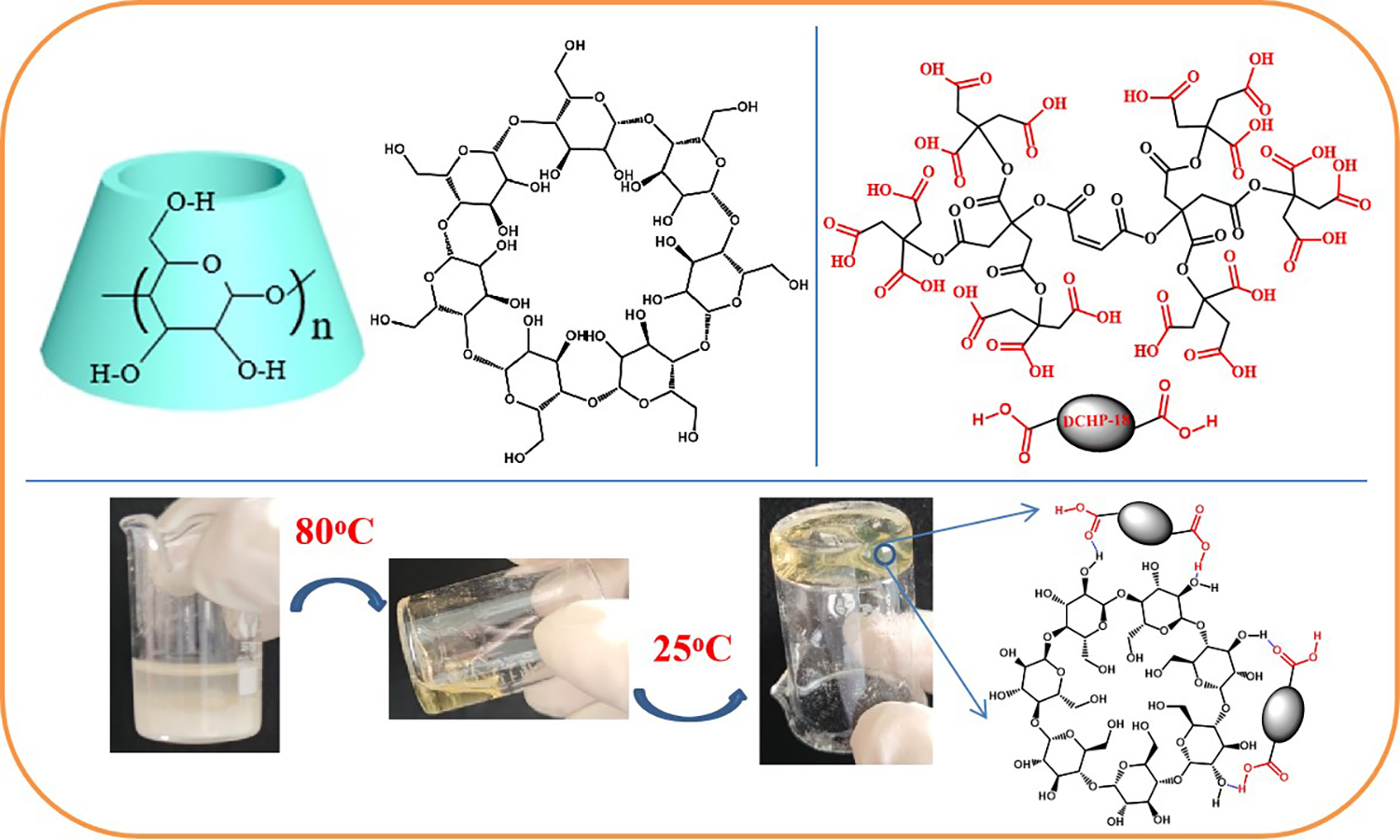
Adhesives have been widely applied in various fields. However, most industrial adhesives contain organic solvents, which are toxic and difficult to clean after use. Importantly, most adhesives are used in a relatively small temperature range (0–50 °C), which is due to the fragmentation of the morphology and properties of supramolecular polymer gels at low temperatures. Most of the supramolecular adhesives need to be heated during the bonding process, which cannot meet the requirements of low-temperature adhesion in daily life and industry. In this study, based on the concept of using deep eutectic solvents (DESs) as a platform, cyclodextrin (β-CD) and carboxyl terminated hyperbranched polyester (DCHP) were used to prepare solvent-free supramolecular polymer gel (CD-DCHP), which was non-toxic and easy to clean. Due to the joint action of deep eutectic solvent and supramolecular polymer structure, CD-DCHP showed excellent mechanical properties, super strong adhesion (up to 4.55 MPa on glass surface), and excellent low-temperature performance (up to 1.64 MPa at –60 °C). This supramolecular polymer gel also showed excellent tolerance to acids, alkalis, and various organic solvents. It greatly expanded the application of DES and provided a new approach to the development of supramolecular adhesives.
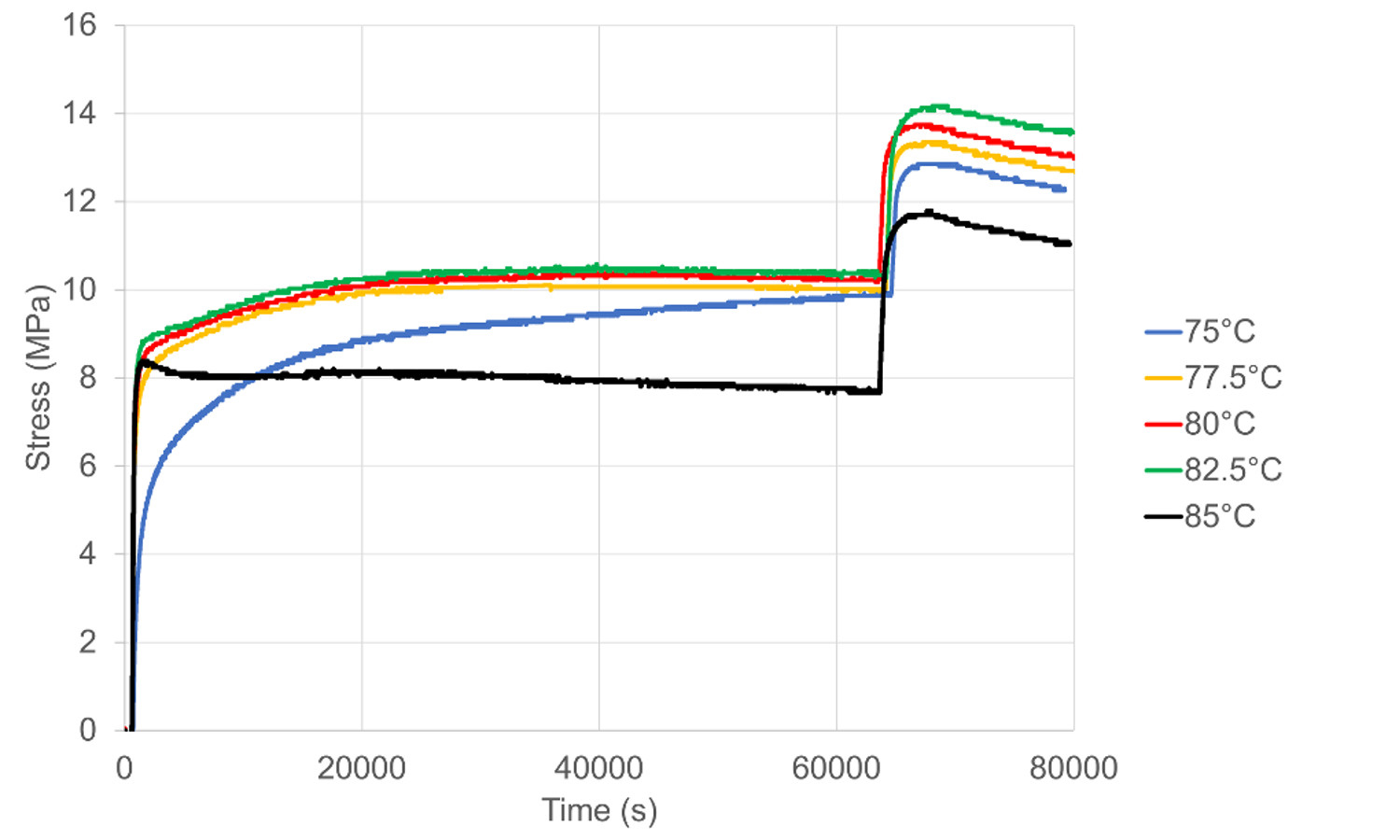
We have performed shrinkage restraint force measurements on three shape memory polymers of polyethylene terephthalate (PET), polymethyl methacrylate (PMMA) and a blend of the two at a range of temperatures. Observations are made of the change in stress during temperature rise, hold and cooling. All materials show an increase in stress during rise and hold, but on cooling, the three materials behave differently; the PET shows a drop in stress, the PMMA a rise and the blend a much smaller rise. This behaviour correlates with the reversible thermal dimensional change below the shrinkage threshold temperature; the expansion coefficients are negative for PET, positive for PMMA and positive at a lower order of magnitude for the blend. We model the behaviour by supposing that the shrinkage forces are created by prestressed strains effective at long range within a matrix of shorter chains effective at short range. The total stress is the sum of the shrinkage stress and the thermal stress in the matrix. The drops in stress on cooling are modelled using an elastic analysis based on measured elastic moduli and thermal expansion coefficients. For the blend, downward jumps in temperature produce small transient increases in the total stress, leaving it effectively unchanged. This phenomenon and the results of the elastic model for the stress drops imply that the shrinkage stress from the long-range chain network is largely unaffected by the temperature change and so is not entropic.
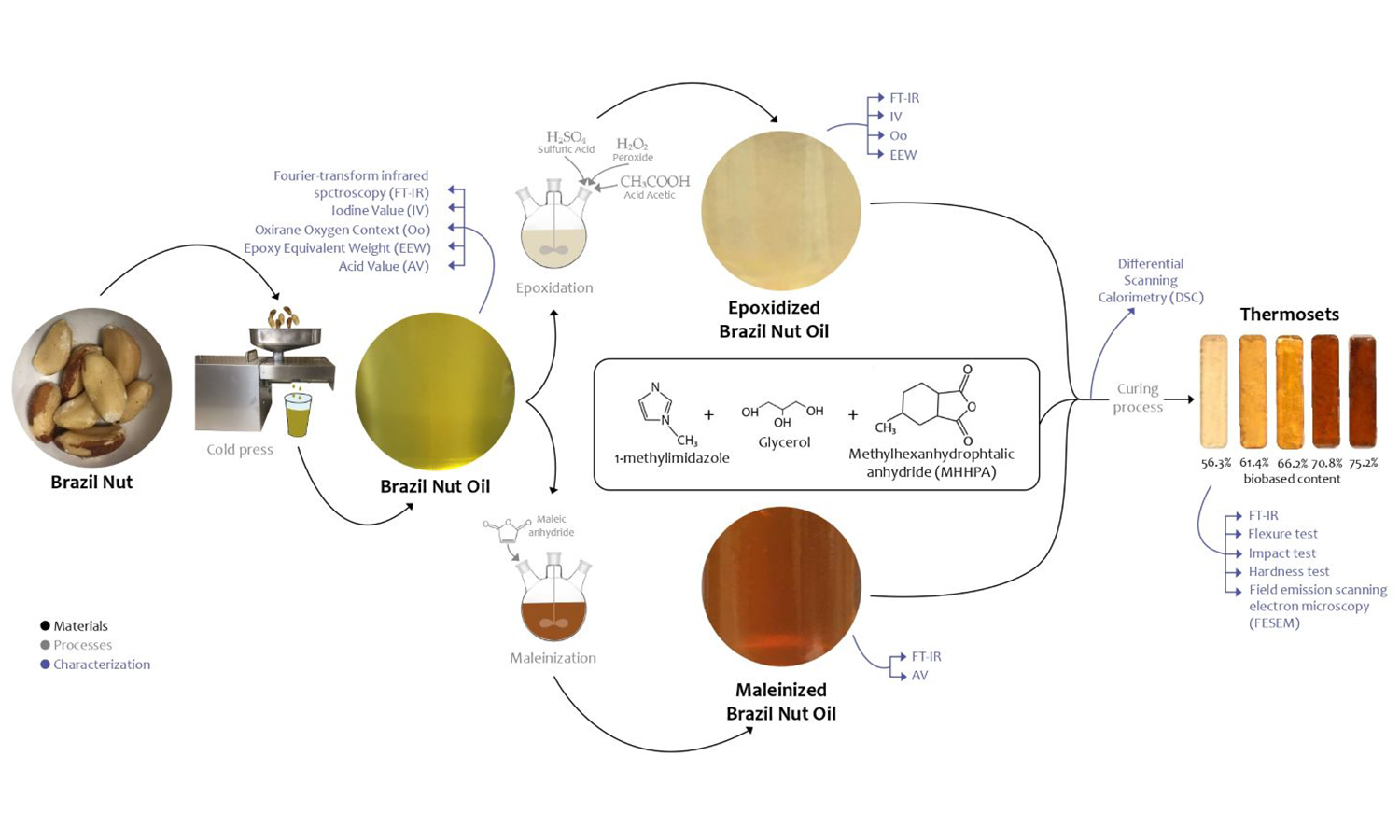
The present work describes, for the first time, the chemical modification and characterization of epoxy resins derived from Brazil nut oil (BNO). The development of a bio-based resin has been carried out by chemically modifying BNO, resulting in epoxidized Brazil nut oil (EBNO). Moreover, through a maleinization process, a crosslinker based on maleinized Brazil nut oil (MBNO) has been employed. Analysis using techniques like iodine value (IV) and oxirane oxygen content (Oo) indicate successful chemical modification. FTIR analysis confirms the presence of reactive functional groups in both EBNO and MBNO, which are responsible for resin crosslinking. Mechanical testing of the different bio-resins developed shows that, in the cured resins, flexural strength and modulus decrease with increasing MBNO content, indicating improved flexibility due to the introduction of MBNO as a crosslinker. Thermal analysis using differential scanning calorimetry (DSC) reveals exothermic peaks corresponding to resin curing appearing at progressively lower temperatures. Therefore, this study successfully modifies BNO and demonstrates its effectiveness as a precursor to develop bio-based epoxy resins. The resulting resins exhibit improved ductile and thermal properties, making them suitable for various applications requiring low mechanical stresses.
Correlation between solution relative viscosity and the microstructural properties of the poly(3-hydroxybutyrate-co-3-hydroxyvalerate) – PHBV solution blow spun mats
Lucas Gomes Rabello, Maira Rievrs Nogueira Alvares, Roberto Carlos da Conceição Ribeiro, Paula Mendes Jardim, Rossana Mara da Silva Moreira Thiré
Vol. 17., No.12., Pages 1239-1256, 2023
DOI: 10.3144/expresspolymlett.2023.94
Vol. 17., No.12., Pages 1239-1256, 2023
DOI: 10.3144/expresspolymlett.2023.94
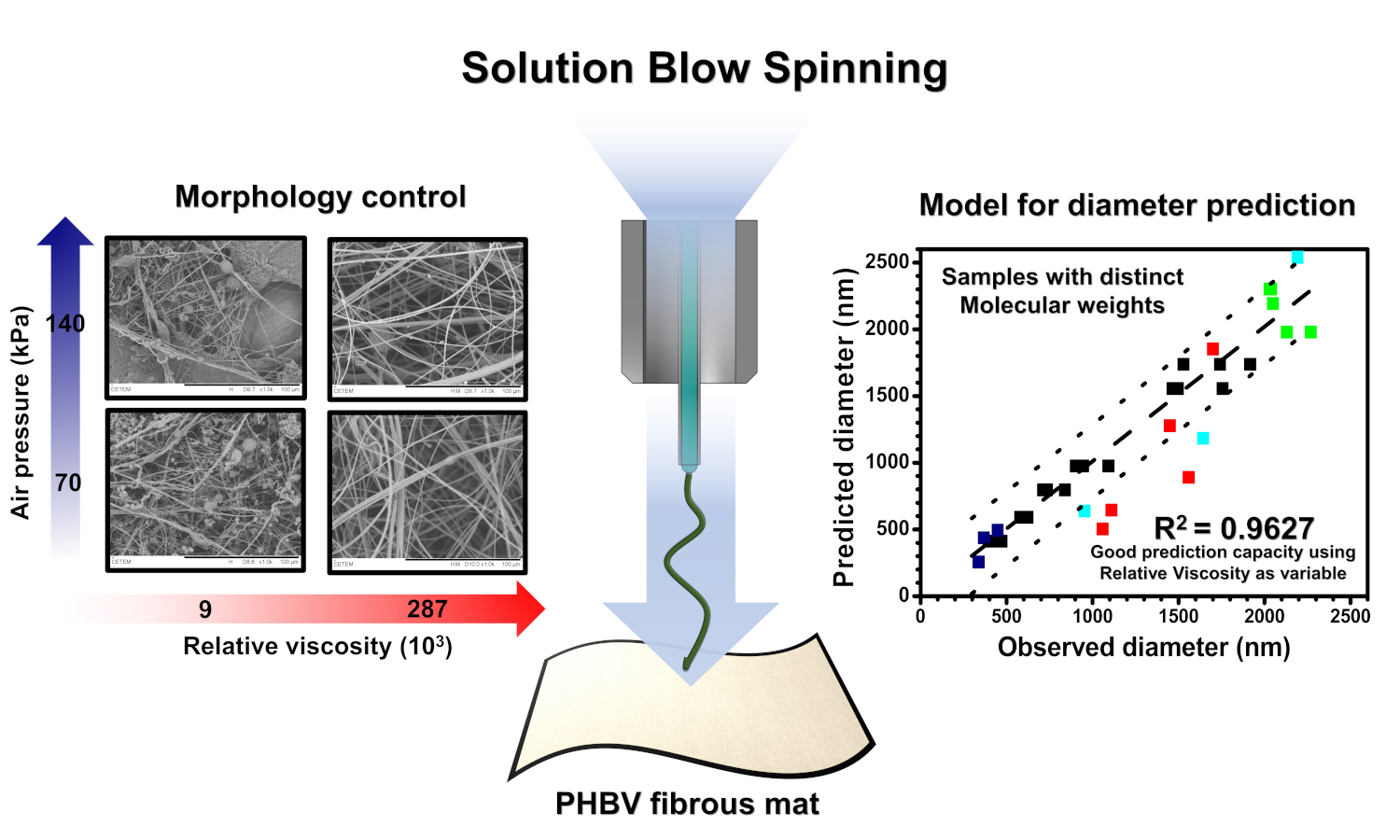
In this study, we produced poly(3-hydroxybutyrate-co-3-hydroxyvalerate) (PHBV) fibrous mats via solution blow spinning (SBS) and evaluated their microstructural properties. We propose here the utilization of the theoretical estimative of relative viscosity (RV) as an independent variable in a statistical design of experiments (DoE) to account for the polymer’s molecular weight (Mw) effect on the determination of the diameter and morphology of the fibers. The RV of the solution (42.3・103–287.4・103) and air pressure – AP (70–140 kPa) were varied. The analysis of variance (ANOVA) indicated that the increase in RV favored an increment in fiber size and resistance to alignment. Higher AP produced aligned and thinner fibers with higher crystallinity. The spinnability regions were determined based on the estimated RV of the PHBV solutions and the AP levels. Fibers were formed at 70 kPa from solutions with RV ranging from 10・103 to 106, while at 140 kPa, from 42・103 to 106. Nanofibers were produced from less viscous solutions (RV = 42.3・103–124.4・103), while microfibers were produced from solutions with RV = 287.4・103. The developed ANOVA model predicted with good accuracy (R2adj. = 0.96) the average diameter of the PHBV fibers produced from the SBS technique using polymers with distinct Mw values, including those available in the literature data.
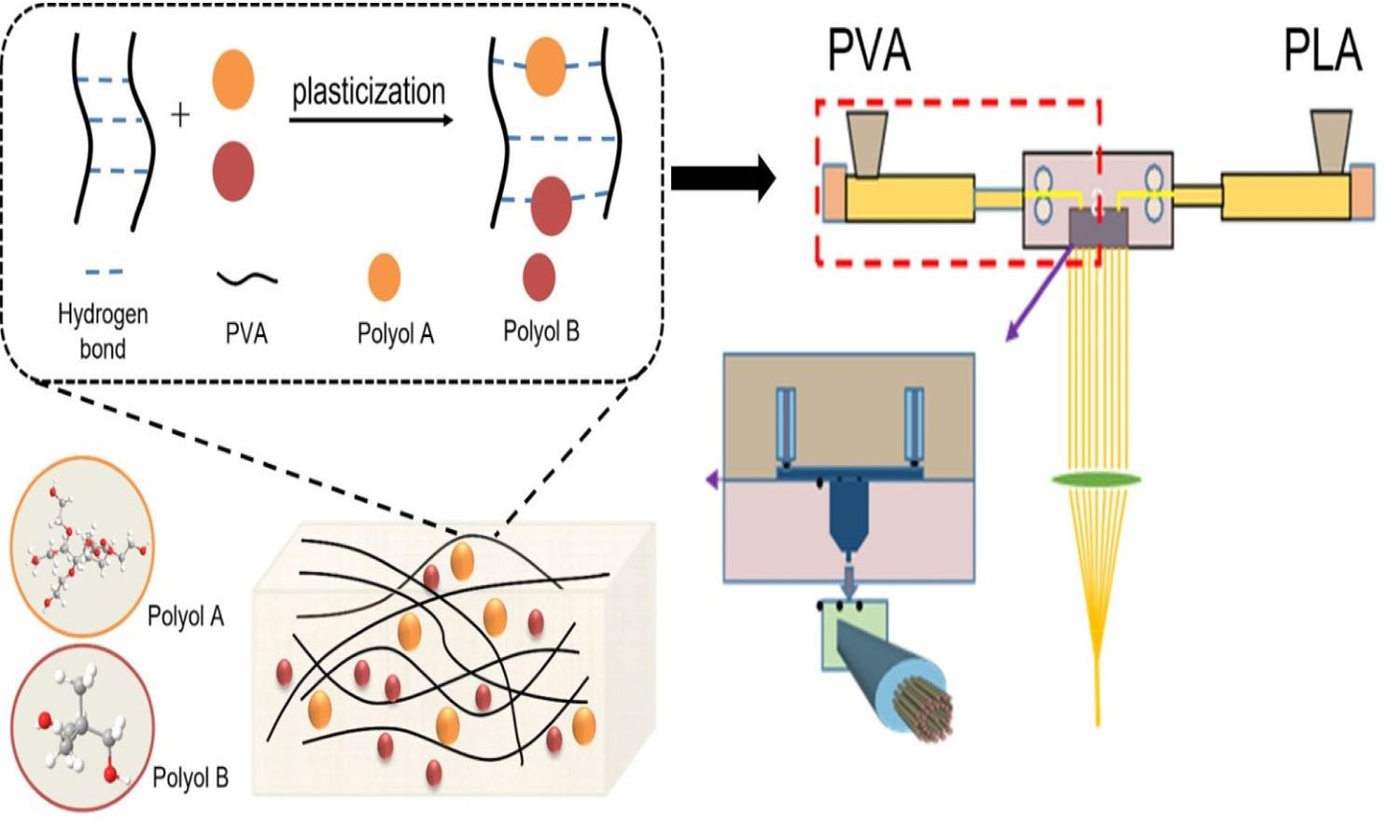
Polyvinyl alcohol (PVA) is a type of water-soluble polymer that is a candidate for ‘sea’ polymer to prepare seaisland fibers in industry. In this study, two types of polyols (sorbitol ether and neopentyl glycol) were mixed in different proportions and used to prepare thermoplastic PVA (TPVA) as a compound plasticizer. When the plasticizer was introduced, a blue shift occurred on the characteristic band of hydroxyl in the Fourier transform infrared spectroscopy (FTIR) pattern. The melting temperature (Tm) of the corresponding TPVA decreased, and their initial decomposition temperature (Td) increased to some degree. The addition of compound plasticizer made PVA show an improved Td up to 273.1 °C. Moreover, it led to an increment of the melting flow index and improved the resistance to oxidation at a temperature higher than 200°C. The increased spinnability enabled TPVA in PVA-PLA sea-island fiber making. PLA superfine fibers were obtained after splitting in water at 95 °C for just 3 min.
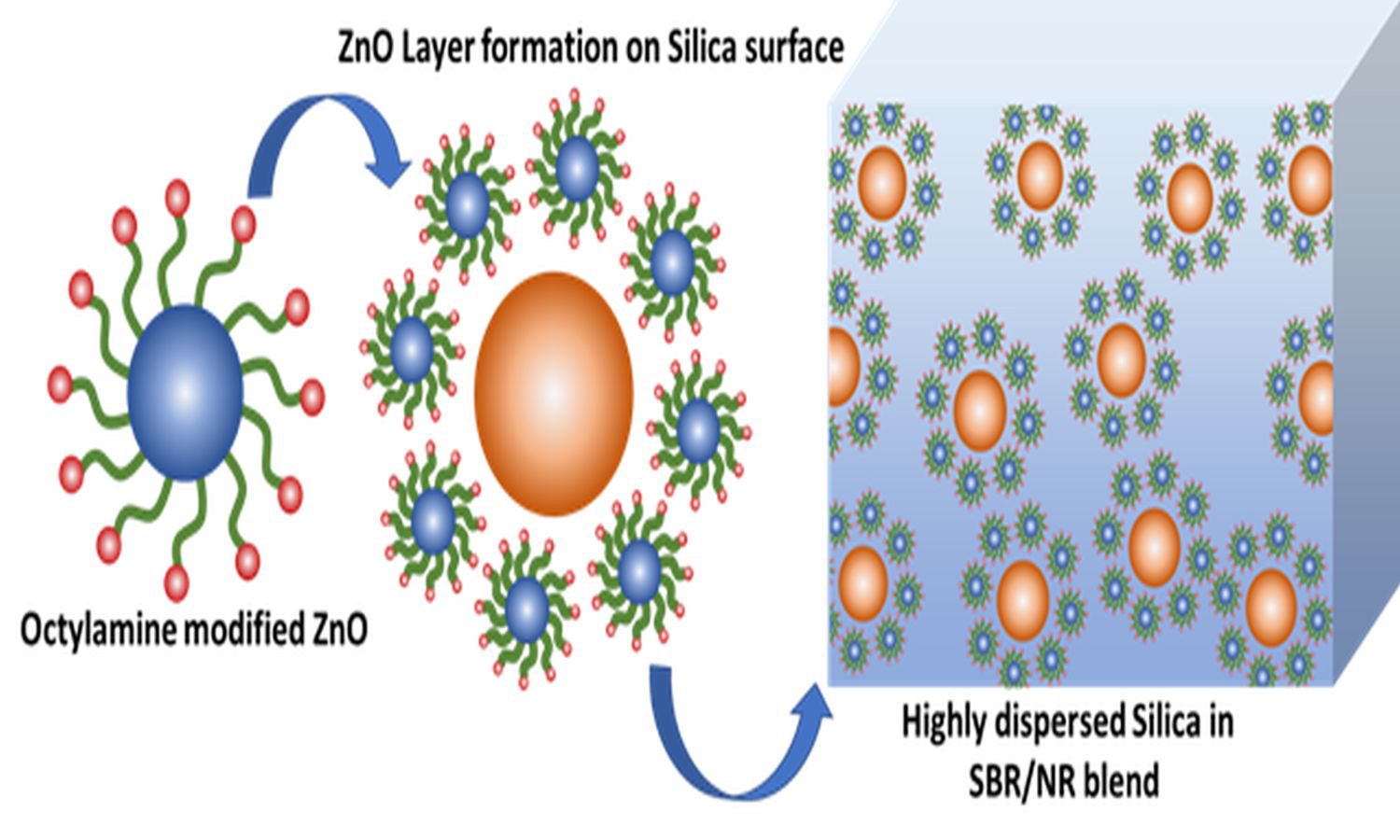
The global concern over zinc leaching into aquatic ecosystems has led researchers to seek ways to reduce zinc oxide (ZnO) content in rubber products. Conventional microsized ZnO, commonly used in the rubber industry, poses dispersion challenges due to its hydrophilic nature and micron size within the hydrophobic rubber matrix. Therefore, higher amounts of ZnO are added, elevating the risk to aquatic life. A promising alternative involves using highly dispersible ZnO with active zinc (Zn) centers instead of conventional ZnO. Another approach includes incorporating ZnO-anchored silica particles into the rubber matrix, which requires additional ex-situ fabrication. This study presents an innovative method where ZnO-anchored silica is generated in situ during the blending of styrene-butadiene rubber/natural rubber (SBR/NR). The study also evaluates the effectiveness of active, nano-sized, and octylamine-modified ZnO as activators compared to conventional ZnO, by introducing silica filler, octylamine-modified and high surface area ZnO anchor onto the silica surface, forming Si–O–Zn covalent bonds. This protective layer reduces filler aggregation and the Payne effect. Even with 60% less usage, these activators in the SBR/NR blend significantly enhance tensile strength (31.27%) and elongation at break (49.13%) compared to conventional ZnO. These results point towards the possibility of a cost-effective and sustainable replacement for conventional ZnO.


